Introduction: When Innovation Is Public, But Ownership Is Private
The COVID-19 pandemic forced governments across the world to fund innovation at unprecedented scale. In India, vaccines like Covaxin became symbols of scientific capability, public resilience, and national self-reliance.
Yet behind the success narrative lies a quieter, more uncomfortable question:
When public money de-risks innovation, who should own the resulting intellectual property?
The Covaxin episode brings this question into sharp focus — not as an allegation, but as a policy lesson.
Covaxin’s Development: A Public–Private Collaboration
Covaxin was not developed in isolation by a private entity.
Key public contributions included:
- ICMR’s scientific collaboration
- NIV Pune’s provision of the SARS-CoV-2 strain
- Use of public research infrastructure
- Regulatory facilitation during an emergency
This collaboration significantly de-risked research and development, shortening timelines and reducing uncertainty for the private partner.
From a policy standpoint, this matters.
The Patent Controversy: Where Transparency Slipped
The controversy did not arise because a patent was filed.
It arose because:
- initial patent filings excluded ICMR as a co-applicant
- inventorship and ownership details were unclear
- licensing and revenue-sharing terms were not publicly disclosed
This created a perception fair or not that public contribution was legally sidelined, even if later corrected.
In high-trust sectors like public health, perception itself is consequential.
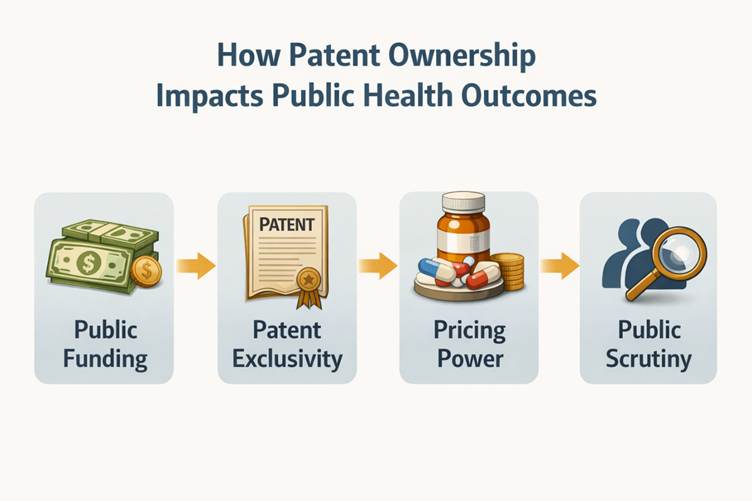
Pricing, Access, and Public Interest
Another flashpoint was pricing.
Questions raised included:
- Should vaccines developed with public funding carry differential pricing?
- Were states and private hospitals charged equitably?
- Did public funding translate into public leverage?
These are not ideological questions.
They are contractual and policy design questions.
The WTO Paradox: Global Advocacy vs Domestic Practice
Internationally, India supported discussions around:
- IP waivers
- equitable vaccine access
- global public health considerations
Domestically, however, the Covaxin episode revealed limited disclosure of IP arrangements.
This divergence exposed a policy gap:
advocating flexibility globally while practicing opacity domestically.
What This Episode Really Exposes
The Covaxin case is not about blame.
It is about system design.
It highlights:
- absence of standard IP clauses in public funding contracts
- lack of default joint ownership norms
- weak public disclosure mechanisms
- no clear march-in or access safeguards
These gaps are structural, not incidental.

Learnings for Future Publicly Funded Innovation
A mature innovation ecosystem requires:
- clear IP ownership rules at funding stage
- transparent licensing terms
- pricing safeguards linked to public funding
- retained government rights in emergencies
Countries that ignore this risk:
- public distrust
- policy backlash
- weakened innovation legitimacy
A Policy Roadmap Going Forward
India must institutionalise:
- mandatory IP disclosure norms for public-private R&D
- default joint ownership in publicly funded health innovations
- compulsory licensing triggers linked to access
- parliamentary oversight of major health IP assets
This protects innovation and credibility.
Conclusion: Innovation Needs Trust, Not Just Patents
Patents are legitimate tools of innovation.
Public funding is a legitimate policy choice.
But when the two intersect, transparency is not optional.
The Covaxin episode should be remembered not as a controversy, but as a turning point — an opportunity to align India’s innovation policy with public accountability.
To understand how intellectual property, public policy, and innovation intersect in India, explore structured learning programs at the Indian Institute of Patent and Trademark (IIPTA).
