“Why PERA 2025 Is a Game-Changer for Innovators”
Intellectual Property Rights, despite of being recognised globally it protects the rights domestically means, if a researcher works energy storage device to store solar energy and applies to patent then he gets the protection in India not in USA unless he applies for the same USPTO. Like other aspects Patents are also governed by the act of law which is enacted by the respective territorial government, The law which has enacted would be amended and modified according to the demand of the situation and time to address the requirements of stakeholders. As we are aware of the fact that Indian Patent Law has amended from time to time where major amendments are like 1999 , 2000 and 2005 and recently Jan Vishwas Act 2023 and Amendment to Rules 2024 which brought considerable flexibilities to ease the filing process and some strict aspects like increasing fees for condonation of delay charging fee for Pre-grant opposition which is a check and balance to take the obstacles created by competitors with malefice intensions.
In the same we are going to analyze one of the Reforms in US, The Patent Eligibility Restoration Act (PERA) 2025. The Patent Eligibility Restoration Act (PERA) of 2025 is a significant legislative proposal aimed at reforming the U.S. patent system by clarifying and expanding the scope of patent-eligible subject matter. PERA seeks to address longstanding ambiguities stemming from judicial interpretations of Section 101 of the U.S. Patent Act.

Due to a series of judicial decisions beginning in 2012—both from the U.S. Supreme Court and the Court of Appeals for the Federal Circuit—patent eligibility law in the United States has become confused, inconsistent, and unclear. The lack of a clear, consistent, repeatable test for what inventions possess the basic threshold characteristics necessary to obtain a patent there has been well-documented and wide-ranging negative impacts within the innovation and investment communities, which has led to unpredictable business outcomes and a stagnation of innovation in certain high-tech industries.
Roadmap of PERA 2025:
The Patent Eligibility Restoration Act of 2025 (PERA 2025) is a bipartisan legislative effort in the United States Congress aimed at reforming patent eligibility law under 35 U.S.C. § 101 to restore clarity, predictability, and global competitiveness to the U.S. patent system.
PERA 2025 would restore patent eligibility to important inventions critical to the growth of the U.S. economy, and critical to delivering on the goal for the United States to dominate AI Landscape. The bill would accomplish the intention and also prevent the patenting of mere ideas, which are already exists in nature, and social and cultural content that virtually everyone agrees is beyond the scope of the patent system.
PERA 2025 is slightly different than the bill introduced in 2023, at least relating to human genes. PERA 2025 still says that unmodified human genes as they exist in the human body are not patent eligible, but prior versions of the bill said that isolation of genes was considered a modification. PERA 2025, however, leaves out the world “isolated” and says that “a human gene shall not be considered to be unmodified if that human gene is purified, enriched, or otherwise altered by human activity; or otherwise employed in a useful invention or discovery.”
Why PERA 2025 Matters
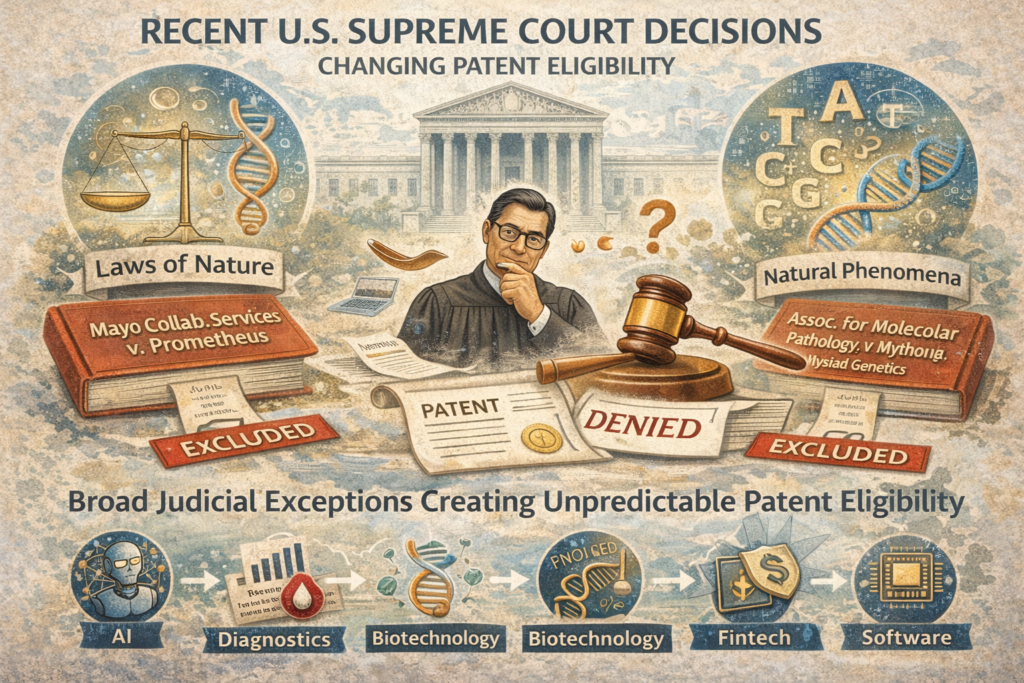
Over the past decade, several U.S. Supreme Court decisions — most notably Mayo Collaborative Services v. Prometheus, Alice Corp. v. CLS Bank, and Assoc. for Molecular Pathology v. Myriad Genetics — created broad judicial exceptions to patent eligibility for “abstract ideas,” “laws of nature,” and “natural phenomena.” These exceptions have made patent eligibility unpredictable, especially for innovation in AI, diagnostics, biotechnology, fintech, and software.
Key Dimensions of PERA 2025:
PERA aims to amend Section 101 of the U.S. Patent Act to provide clearer guidelines on patent eligibility. The bill’s main provisions include:
- Elimination of Judicial Exceptions: PERA proposes to remove all judicially created exceptions to patent eligibility, such as abstract ideas, laws of nature, and natural phenomena.
- Defined Ineligible Subject Matter: The bill specifies certain categories that would remain ineligible for patent protection:
- Mathematical formulas not integrated into a practical application.
- Mental processes performed solely in the human mind.
- Unmodified human genes as they exist in the human body.
- Unmodified natural materials as they exist in nature.
- Processes that are substantially economic, financial, business, social, cultural, or artistic in nature.
- Clarification on Machine-Implemented Processes: Processes that cannot be practically performed without the use of a machine (including a computer) or manufacture would be considered patent-eligible.
- Separation from Other Patent Requirements: PERA emphasizes that patent eligibility should be determined independently of other patentability criteria such as novelty (Section 102), non-obviousness (Section 103), and disclosure requirements (Section 112).
Support and Opposition
Supporters of PERA argue that the bill would:
- Restore clarity and predictability to patent law, encouraging innovation and investment.
- Align U.S. patent eligibility standards more closely with those of other countries, enhancing global competitiveness.
- Provide stronger protection for advancements in emerging technologies like artificial intelligence, quantum computing, and personalized medicine.
Opponents, including some public interest groups and scholars, express concerns that:
- The elimination of judicial exceptions could lead to the patenting of fundamental scientific principles and natural phenomena, potentially hindering research and access to essential technologies.
- Allowing patents on unmodified human genes and naturally occurring associations between genes and diseases could increase costs and limit access to genetic testing and diagnostics.
Legislative Outlook
As of May 2025, PERA has garnered bipartisan support and is under consideration in Congress. While the bill aims to address critical issues in patent law, its passage will depend on ongoing debates and negotiations among lawmakers, stakeholders, and interest groups. The outcome will significantly impact the future landscape of innovation and patent protection in the United States.

What Innovators & R&D Teams Should Learn from This Case Before filing Conventional Application in general and USPTO filing particularly?
Basic Dimensions
Disclose Completely
Any patent application MUST include:
- Genetic sequence information
- Methodology
- Lab protocols
- Culture conditions
- Experimental results
- Deposit details (if needed)
Since India and other global stakeholders’ demands high scientific detail, not summaries.
Draft Claims Smartly not broadly which is an essential aspect across the patent landscape:
Smart claims are those when they:
- Match the description
- Are precise
- Highlight inventive steps
- Exclude natural organisms
A well-drafted claim is proportional to Higher chance of grant.
Understand the patentability and non-patentability criteria of the designated country.
In Indian scenario Section 3 restricts
Common rejection grounds:
- Section 3(c) – natural organisms
- Section 3(d) – known variants
- Section 3(i) – diagnostic & therapeutic methods
- Section 3(j) – plants, animals, seeds
- Section 3 (k) – mathematical model and algorithms
- Section 3 (m)- mental act
- Section 3 (n) – presentation of information
If your invention falls here there is a for need expert drafting to overcome objections as well as rejection.
Impact on Biotech Startups & Researchers
This amendment signals that:
- Indian Patent law is in consonance according to international standards and also operating according to the framework of international guideline strategies.
- BUT only when applications are drafted with precision and scientific depth then only it meets the international standards.
For startups, researchers, and companies, the key considerations are as follows:
- Needful preparation of detailed, high-quality patent specifications.
- Need to work with experts who understand both science as well as patent law.
- Need to avoid broad, vague, or incomplete disclosures.
How IIPTA Helps Biotech Innovators & Students
At IIPTA (Indian Institute of Patent & Trademark),
we help:
- Innovators
- R&D teams
- Startups
- Students
- Biotech professionals
In understanding patent drafting, Section 3 compliance, and global patent trends.
We offer:
- Patent drafting training
- Patent agent exam preparation
- Real case-law analysis
- One-to-one expert guidance
- Hands-on practice with live international cases
Conclusion : Despite of being domestic rights intellectual property in general and patent in particular are granted according to global standards and it is essential for the IPR professionals to upgrade the execution service to global landscape standards with updating of International dynamics.
